DNA and RNA - Transcription
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#RNAtranscription #mRNA #RNA
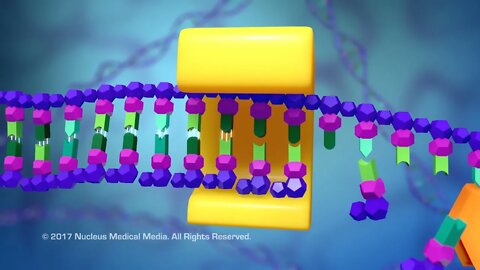
SCIENCE ANIMATION TRANSCRIPT: Now, that we've covered DNA replication, let's talk about Transcription. The first thing you need to know is that transcription has nothing to do with cell replication processes, such as DNA replication, mitosis, or cell division. So what is transcription and why is it necessary? Well, transcription is the first step in the process of using the genetic code in DNA to synthesize or build all the different proteins in your body. One problem with synthesizing these proteins is that the instructions for making them are in the DNA, which is located inside the nucleus. But the place proteins are always made is outside the nucleus, either in the ribosomes floating in the cytoplasm, or in the ribosomes embedded in the rough endoplasmic reticulum. So, how does the genetic code for synthesizing proteins get from the DNA to the ribosome? DNA uses a messenger called messenger RNA or mRNA to carry the genetic code from the nucleus to the ribosome. The process of building this messenger RNA is called Transcription. Now, let's see how transcription happens. Transcription begins when an enzyme called RNA polymerase attaches to a segment of DNA called a gene. A gene contains the code to build a specific protein, which is a macromolecule made up of a sequence of amino acids in a specific order. And within a gene, the specific order of nitrogenous bases dictates the order of amino acids that will make up the protein. Each group of three consecutive bases in the gene is actually a code for a particular amino acid. As a result, each group is referred to as a codon. RNA polymerase causes a particular area of the DNA helix to unwind and separate into two strands. One of the strands, often called the template strand, is the side of DNA that is read or transcribed by the messenger RNA. The other strand of DNA, often called the non-template strand, isn't transcribed by the messenger RNA. So how are DNA instructions transcribed into messenger RNA? Well, using the template strand as a guide, RNA polymerase uses the base pair rule to assemble free nucleotides in the nucleus into a complementary strand of RNA. For example, RNA polymerase reads the DNA base thymine on the template strand then binds it to a free nucleotide containing adenine. This process continues with cytosine binding to guanine and guanine binding with cytosine. Remember though, RNA will never contain thymine. So, whenever RNA polymerase sees adenine on the DNA template strand, it pairs adenine with uracil. By using the template strand of DNA as a guide, the genetic code from the non-template strand of DNA has actually been transcribed into messenger RNA. When transcription is complete, the messenger RNA, which is small enough to fit through a nuclear pore, takes the genetic code out of the nucleus to the ribosome, the site of protein synthesis. The process of actually building the protein at the ribosome is called Translation, which we'll cover in a separate video. To summarize, transcription is the process of transcribing or copying the genetic code for building a protein into messenger RNA. A gene is a segment of DNA containing the instructions or code for building a protein. A codon is a group of three consecutive nitrogenous bases in a gene containing the code for a specific amino acid in a protein. RNA polymerase unwinds the strands of DNA in a gene. The template DNA strand contains the complementary bases that need to be read to generate messenger RNA. The base pair rule is followed when assembling messenger RNA. Messenger RNA is actually a copy of the DNA non-template strand with uracil substituted for thymine.
NSV16039
212
views
DNA and RNA - DNA Replication
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#DNAreplication #DNAmolecule #DNA
SCIENCE ANIMATION TRANSCRIPT: Let's take a look at DNA replication, the process in which DNA copies itself. Why does DNA need to copy itself? Well, before a cell divides during mitosis, it must make a copy of its original DNA. This ensures that both resulting daughter cells will have DNA that is identical to the original cell's DNA. So at what point in the cell cycle does DNA replication happen? DNA is copied or replicated during the S phase of the cell cycle. A good way to remember this is that S stands for synthesis, which means to make. Now, let's see how DNA replicates during the S phase. If we zoom into the nucleus, we can see the DNA molecules two strands or sides twisted together in the classic double-helix formation. The two strands are connected by hydrogen bonds between their nitrogenous bases. The first step of replication begins with an enzyme called DNA helicase. The suffix A-S-E or ASE tells you right away that helicase is an enzyme. Helicase unwinds and then separates the two sides of the DNA molecule by breaking the hydrogen bonds between their nitrogenous bases. Each separate half of the DNA can now serve as a template or pattern for the creation of a new strand of DNA. This separation exposes all of the nitrogenous bases on both sides of the DNA to the environment within the nucleus where free nucleotides are present. This brings us to the second step in DNA replication, the creation of two new identical DNA strands. An enzyme that assists in this process is called DNA polymerase. It adds free nucleotides available in the nucleus to the original template strands. DNA polymerase does this by creating new hydrogen bonds between the available nitrogenous bases of the free nucleotides and those on both sides of the original DNA molecule. New nucleotides are added to the template strands following the base pair rule of nitrogenous bases. Remember, in DNA, adenine always bonds with thymine. Thymine always bonds with adenine. Guanine always bonds with cytosine, and cytosine always bonds with guanine. So these newly attached nucleotides form a mirror image or complementary strand on each template strand of the original DNA. As a result of this replication process, two duplicate molecules of DNA are produced from the original DNA molecule. With DNA replication now completed, the cell is ready to begin mitosis. We will cover mitosis in another video. In summary, DNA replication is a process in which DNA duplicates itself, making an identical copy. Replication occurs during the S phase of the cell cycle when a somatic cell is preparing to divide. DNA replication is necessary so that the two daughter cells produced after mitosis both have DNA that is identical to each other and identical to the DNA in the original cell. At the beginning of replication, an enzyme called DNA helicase breaks the hydrogen bonds between nitrogenous bases and unwinds the DNA molecule. Each side of the original DNA molecule serves as a template for the creation of a new complementary strand of DNA. With the assistance of DNA polymerase and other enzymes, free nucleotides are added to the template strands following the base pair rule. The result of DNA replication is two identical duplicate DNA molecules from the original DNA molecule. ♪ [music] ♪
NSV16037
83
views
DNA and RNA - Overview of DNA and RNA
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#NucleicAcids #DNA #RNA
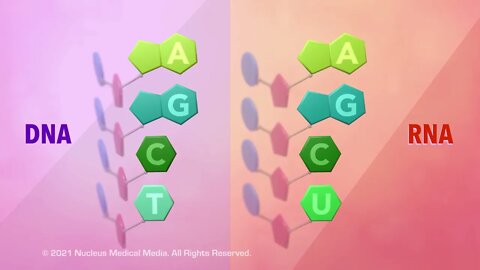
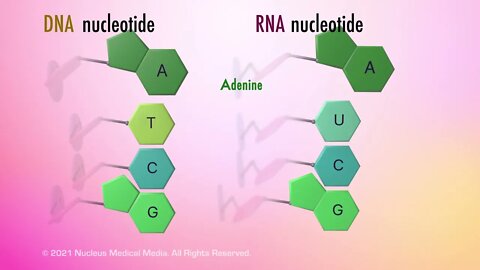
SCIENCE ANIMATION TRANSCRIPT: Today, we're going to be talking about the only two types of nucleic acids that exist, DNA and RNA. DNA stands for deoxyribonucleic acid, and RNA stands for ribonucleic acid. As nucleic acids, both DNA and RNA are long organic macromolecules, which means they are very large molecules that contain the element carbon. Both DNA and RNA are composed of nucleic acid monomers called nucleotides. Nucleotide monomers always contain a phosphate group, a five-carbon sugar, sometimes referred to as a pentose, and a nitrogenous base. But DNA nucleotides have the five-carbon sugar deoxyribose while RNA has a different five-carbon sugar called ribose. The pentose sugar in DNA has one less oxygen than ribose in RNA, which leads to its name, deoxyribose. Let's focus on the nitrogenous bases in DNA for a moment. Each DNA nucleotide contains only one of four possible nitrogenous bases, adenine, guanine, thymine, and cytosine. These four bases are usually abbreviated A, G, T, and C. When nucleotide bases connect through hydrogen bonds, it's important to know that the bases always pair up with the same partners. Adenine only pairs with thymine, and guanine only pairs with cytosine. This is called the base pair rule. Scientists classify nitrogenous bases according to the shape of their molecules. Adenine and guanine both have a double-ring structure containing a six-membered and a five-membered nitrogen-containing ring fused together. These types of ring structures are chemically classified as purines. But thymine and cytosine, both, only have a single six-membered nitrogen-containing ring shape, so they are chemically classified as pyrimidines. Here's a little hint to help you remember this. Pyrimidine has a Y in its name, and so do the bases classified as pyrimidines, thymine and cytosine. Like DNA, an RNA nucleotide may have the nitrogenous bases, adenine, guanine, or cytosine. But unlike DNA, which has the nitrogenous base thymine, RNA has the nitrogenous base uracil instead. This means that any nucleic acid that contains thymine must be a strand of DNA and that any nucleic acid that contains uracil must be a piece of RNA. Like thymine, uracil has a single nitrogen-containing ring. So it would also be classified as a pyrimidine. So what do the nitrogenous bases have to do with building a DNA molecule? Well, each strand of DNA begins to assemble when the phosphate group in one nucleotide bonds with the five-carbon sugar in another nucleotide. The two strands that make up a DNA molecule are linked through hydrogen bonds between their nitrogenous bases. When the spiral DNA molecule is untwisted, these base pairs look like rungs on a ladder. The spiral DNA molecule is described as having a double-helix shape. James Watson and Francis Crick first described DNA's double-stranded helical structure in 1953 after looking at x-ray images of a DNA molecule taken by Rosalind Franklin. Now, let's look at the structure of RNA and see how it compares to the structure of DNA. While DNA is a double-stranded molecule, RNA is always single stranded. In eukaryotic cells, DNA is found only in the nucleus. But RNA can be found in the cytoplasm as well as in the nucleus. DNA controls heredity by containing the code or set of instructions for building the functional and structural proteins that make up your body. And RNA uses these varied instructions to build all the proteins a living organism needs. There are three types of RNA, messenger RNA, referred to as mRNA, ribosomal RNA, referred to as rRNA, and transfer RNA, referred to as tRNA. Messenger RNA starts off in a cell's nucleus. Here, it transcribes or copies the instructions for assembling a protein from a portion of the cell's DNA. Next, the messenger RNA takes these instructions or protein recipe to a ribosome in the cytoplasm. The ribosome, which is made out of ribosomal RNA and proteins, is the site where this recipe is read and translated into a protein. Finally, transfer RNA in the cytoplasm brings specific amino acids to the ribosome so that the particular protein encoded by the recipe can be made. We'll cover more detail about the way DNA and RNA make proteins in another video. To sum up, DNA and RNA are the only two nucleic acids that exist. Both DNA and RNA are built from nucleic acid monomers called nucleotides. Nucleotides always contain a phosphate group, a five-carbon sugar, and a nitrogenous base. Deoxyribose is the sugar in DNA, and ribose is the sugar in RNA. The nitrogenous bases in DNA are adenine, thymine, cytosine, and guanine. The nitrogenous bases in RNA are adenine, uracil, cytosine, and guanine. In DNA, adenine always pairs together with thymine, and cytosine always pairs together with guanine...
NSV16034
104
views
Biology Quiz: True or false?Animal cells have a cell wall.
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Animal cells never have a cell wall.
35
views
Biology Quiz: What structure gives plant cells shape, support and protection?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Plant cells also have a cell wall outside of their cell membranes that shape, support and protect the plant cell.
18
views
Biology Quiz: What makes chloroplasts green?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
It's green because it has a green pigment, called chlorophyll.
21
views
Biology Quiz: In what organelle does photosynthesis take place?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
The chloroplast is where photosynthesis happens.
15
views
Biology Quiz: What is the term for organisms that capture sunlight for energy?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Some organisms, such as plants, that are photoautotrophic — meaning they capture sunlight for energy — have cells with an organelle called a chloroplast.
13
views
Catalysts and Enzymes
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#catalysts #enzymes #ActivationEnergy
SCIENCE ANIMATION TRANSCRIPT: Today, we're going to talk about catalysts and enzymes. To understand enzymes, we need to know that chemical reactions require energy to occur. Recall that chemical reactions break the chemical bonds in reactants and rearrange those bonds to make products. Let's look at an energy diagram to see how energy changes as a chemical reaction progresses. At the start of a reaction, the reactants will have some amount of energy. If the products end up with more energy than the reactants had at the beginning, then this means the reaction absorbed energy from the environment. This is called an endothermic reaction. However, if the products end up with less energy than the reactants, then this means the reaction released energy. This is called an exothermic reaction. Either way, notice the peak in the energy hill just before the products begin to form. This peak represents the minimum energy the reactants required for the reaction to take place. The reactants need this energy to break their chemical bonds so that different bonds can be formed to make new products. This minimum amount of energy required for a reaction to take place is called activation energy. If the activation energy isn't reached, no reaction takes place. Sometimes, scientists need to make a reaction happen faster. If you want to speed up a reaction, you could increase the energy of the reactants by increasing their temperature, or you could add something called a catalyst. A catalyst is a substance that catalyzes or speeds up a chemical reaction without it being changed or used up by the reaction. Catalysts work by lowering the activation energy needed for the reaction to take place. By requiring less energy, catalysts make reactions happen faster and more efficiently because more of the reacting particles are likely to have sufficient energy or activation energy. And the higher the catalyst concentration, the faster the reaction takes place. Since catalysts aren't changed or used up by a reaction, they're available to catalyze more reactants. Chemical reactions are done in labs, but many chemical reactions also happen constantly inside your body. In living organisms, many reactions require catalysts. Biological catalysts are called enzymes. An enzyme is always made up of proteins. Proteins are one of the four types of organic compounds. We'll discuss them in more detail separately. Enzymes make life possible by lowering the amount of activation energy needed and, therefore, speed up reactions that would otherwise take too long. You need enzymes to stay alive. A reactant acted upon by an enzyme is called a substrate. Each enzyme has a unique active site that only a certain substrate can attach to, like a very specific puzzle piece. When attached, they form what is called an enzyme-substrate complex. This fit between an enzyme and substrate is so specific that it's often compared to a lock and key. When the substrate attaches to the enzyme, the enzyme lowers the activation energy needed to break the bonds in the substrate. Once the bonds break in the substrate, it separates into multiple components called products, which leave the active site. Afterward, the enzyme is still available to participate in another reaction with the same type of substrate. So, what factors affect how well an enzyme works? Well, enzymes work best around particular pH levels and certain temperatures, and the presence or absence of other chemicals can also impact their effectiveness. Not surprisingly, humans and other organisms have adapted to use enzymes that are most effective at the organism's natural temperature and pH values. To sum up, chemical reactions require energy to break the reactants' chemical bonds. The minimum amount of energy required for a reaction to happen is called the activation energy. Catalysts speed up reactions by lowering the activation energy. Catalysts aren't used up by chemical reactions. Biological catalysts are called enzymes. Enzymes are made of proteins. Enzymes act on reactants called substrates. Substrates bind to unique locations called active sites on enzymes. And factors that affect how well an enzyme works include pH, temperature, and other chemicals.
NSV16023
43
views
Can You Answer ALL 28 Questions on Cell Biology? (Everyone misses #22)
If you are an educator at K-12, college, university or medical school, you may download 8 FREE medical animations from Nucleus by signing up for a free trial: http://nmal.nucleusmedicalmedia.com/free-trial-membership-a
23
views
Biology Quiz: What are the 2 components of the cytoskeleton?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
The cytoskeleton includes the threadlike microfilaments, which are made of protein, and microtubules, which are thin, hollow tubes.
14
views
Biology Quiz: What is the main function of the cytoskeleton?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Meanwhile, the cell maintains its shape through a cytoskeleton.
14
views
Nucleic Acids
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#NucleicAcids #DNA #RNA
SCIENCE ANIMATION TRANSCRIPT: The final organic macromolecule we'll cover is nucleic acids. A nucleic acid is an organic macromolecule, which means it's a large organic compound made from thousands or hundreds of thousands of smaller molecules. There are two types of nucleic acids, deoxyribonucleic acid or DNA and ribonucleic acid or RNA. Notice that both terms actually contain the words nucleic acid in them. In living organisms, DNA is the main component of chromatin, which will condense into the familiar chromosome shape prior to cell division. The DNA in chromosomes is organized in a specific order that makes up an organism's genes. Genes contain the directions for every function, trait, and activity in a living organism. These activities include growth, reproduction, and especially, heredity, which means the ability to pass on genes to offspring. The other type of nucleic acid, RNA, is integrally involved in building specific proteins by assembling their amino acids in the correct order. Proteins are responsible for all cellular functions in living organisms. So what are nucleic acids made of? Of course, as an organic macromolecule, nucleic acids contain the element carbon. Like proteins, nucleic acids also contain hydrogen, oxygen, and nitrogen. But nucleic acids also contain the element, phosphorous. So what is the structure of nucleic acids? Well, both DNA and RNA are made up of monomers called nucleotides. All nucleotides contain three compounds. The first compound is a phosphate group which contains phosphorous. The second compound is a five-carbon sugar. All five-carbon sugars can be called a pentose. A good way to remember this is that pent means five and the suffix, -ose, means sugar. The specific pentose sugar in RNA is called ribose. The pentose sugar in DNA has one less oxygen than ribose in RNA, which leads to its name, deoxyribose. The third compound in a nucleic acid nucleotide is a weak base that always contains nitrogen. For this reason, this compound is referred to as a nitrogenous base. The nucleotides in DNA contain one of four possible nitrogenous bases. They are called adenine, thymine, cytosine, or guanine. These bases are usually abbreviated as A, T, C, and G. Similarly, the nucleotides in RNA also contain one of four nitrogenous bases, adenine, uracil, cytosine or guanine. They are abbreviated as A, U, C, and G. Notice that both DNA and RNA have adenine, cytosine, and guanine but only DNA has thymine and only RNA has your uracil. So how do nucleotide monomers assemble into nucleic acids? Well, the phosphate group in one nucleotide bonds with the pentose sugar in another nucleotide. The assembled string of nucleotide forms a nucleic acid polymer. Even though both RNA and DNA have alternating sugar and phosphate groups, there are differences in their overall structure. For example, RNA is a single-stranded nucleic acid while DNA is a double-stranded nucleic acid. The two strands in DNA are linked by a hydrogen bond that connects the nitrogenous bases from one strand to the nitrogenous bases from the other strand. In summary, nucleic acids are organic macromolecules. The two types of nucleic acids are DNA and RNA. DNA and RNA are used to transmit hereditary information and to instruct the cell how to properly construct proteins. Nucleic acids contain carbon, hydrogen, oxygen, nitrogen, and phosphorous. Nucleotides are the monomers that bond together to form DNA or RNA. Nucleotides consist of three compounds, a phosphate group, a five-carbon sugar, also known as a pentose, and a nitrogenous base. The nitrogenous bases in DNA are adenine, thymine, cytosine, and guanine. The nitrogenous bases in RNA are adenine, uracil, cytosine, and guanine. The five-carbon sugar in DNA is called deoxyribose while the five-carbon sugar in RNA is called ribose.
NSV16033
79
views
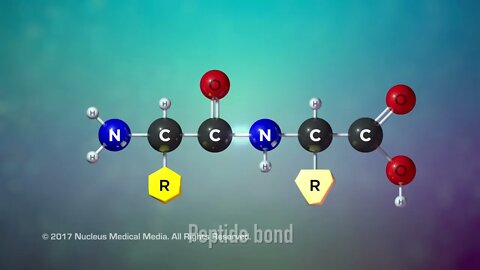
Proteins
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#proteins #AminoacidMolecule #peptides
SCIENCE ANIMATION TRANSCRIPT: So far we've covered two of the organic macromolecules: carbohydrates and lipids. Today, we'll be talking about proteins. Proteins have many uses. For example, many different proteins are used to form the majority of various body tissues including muscle, bone, cartilage, and hair. Other proteins form specialized channels in cell membranes to let certain substances into or out of the cell. Immunoglobulins, also known as antibodies, are made of proteins. They are an important part of the immune system. Antibody proteins label foreign substances in the body so that the immune system can identify and destroy them. And all living organisms use regulatory proteins called enzymes to speed up chemical reactions in the body. So what are proteins made of? Well, all proteins are organic macromolecules, which means they're large molecules containing the element carbon. Like carbohydrates and lipids, proteins also contain hydrogen and oxygen. In addition, proteins also contain the element nitrogen. So what is the structure of proteins? Well, the protein building block, or monomer, is something called an amino acid molecule. Every amino acid has a particular structure. They all have the same amino group on one side and a weak acid called a carboxyl group on the other. But each amino acid also has a unique side chain called an R-group. It's made up of one or more atoms that distinguish one amino acid from another. There are only 20 possible different R-groups, so that means there are only 20 possible different amino acids. A protein is created when amino acids chemically bond to each other. The bonds that connect amino acids to make a protein are called peptide bonds. Small protein molecules are sometimes referred to as protein polymers or peptides. Like interconnecting toy blocks, amino acids can join together to form even longer protein polymers called polypeptides. Changing the order of the amino acid sequence or the length of the polypeptide is what creates countless different proteins. In addition, a protein's final folded shape is critical to the function it performs. Heat or chemicals can change a protein's shape but not its amino acid order. However, the change in a protein's shape can cause the protein to lose its functionality. This is called denaturing a protein. Denaturing is usually not reversible. An example of denaturing a protein happens when you fry an egg. Egg whites are pure protein. If you heat an egg, the protein in the egg white becomes denatured. You can see the result of this denaturing as the egg white changes from a clear-colored liquid to a white-colored solid. Even after cooling, the egg whites' denatured proteins can't change back to their original shape. So the egg white retains its opaque white color and solid form. To sum up, proteins are organic macromolecules containing carbon, hydrogen, oxygen, and nitrogen. Enzymes, most body tissues, and many cells in the immune system are all made up of proteins. Amino acids are the monomers that make up proteins. There are only 20 different amino acids. Small protein polymer molecules are often called peptides. Long chains of amino acids are also a type of protein polymer called polypeptides. Proteins differ in amino acid sequence, polypeptide length, and folded shape. A protein's folded shape is critical to its function. ♪ [music] ♪
NSV16038
39
views
Biology Quiz: Cells needing more energy require more of which organelles?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Cells that need more energy have more mitochondria.
3
views
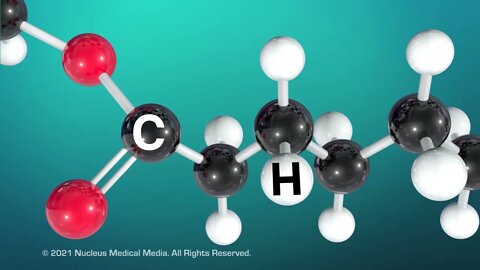
Lipids
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#lipids #fats #steroids
SCIENCE ANIMATION TRANSCRIPT: Today, we're going to talk about lipids. Lipids are an integral part of every cell membrane in every living organism. Looking closer, you can see that lipids are part of the phospholipid membrane that is the boundary of every single cell. Most people think of lipids as fats, such as the fat in your body. Fats provide long-term energy storage and insulation in living organisms. But fats are just one type of lipid. Oils, waxes, and steroids are also types of lipids. Examples of steroids include cholesterol and hormones such as testosterone, which is produced in the testicles, and estrogen, which is produced in the ovaries. A common feature of all lipids is that they don't dissolve in water. So what makes something a lipid? All lipids are organic macromolecules. This means lipids are large molecules containing the element carbon. Lipids also contain hydrogen and oxygen. Organic macromolecules, such as lipids are formed by many units called monomers that are chemically bonded together. In lipids, the typical monomer is something called a fatty acid. A fatty acid contains a chain of carbon atoms attached to each other. Hydrogen atoms are also attached to these carbon atoms. You may recall that carbon can form up to four covalent bonds with other atoms. When each carbon atom forms two single bonds with adjacent carbon atoms and another two single bonds with adjacent hydrogen atoms, we call this fatty acid saturated. This means the fatty acid is saturated with all the hydrogen atoms it can possibly contain. Because of this structure, saturated fatty acids are straight molecules that can pack tightly together. As a result, saturated fats are usually solid at room temperature. Examples of saturated fats include lard and butter. In contrast, unsaturated fats are usually liquid at room temperature. These include things like vegetable oil and olive oil. So why are unsaturated fats liquid at room temperature? Well, unsaturated fatty acids have one or more double covalent bonds between carbon atoms. This means the unsaturated fatty acid has gaps in the hydrogen saturation. The carbon double bonds form kinks in the fatty acid, which prevent the molecules from packing together tightly. As a result, the more loosely packed molecules form a liquid instead of a solid at room temperature. Here's a tip to help you remember which type of fat is solid or liquid. Use the letter S at the beginning of the word saturated for solid at room temperature. Now that we've talked about fatty acids as lipid monomers, what is a lipid polymer? A lipid polymer, called a triglyceride, is formed when three fatty acids bond to a glycerol molecule. A triglyceride is saturated if it contains only saturated fatty acids. And a triglyceride is unsaturated if it contains any unsaturated fatty acids. To sum up, all living organisms use lipids in all of their cell membranes, as well as for long-term energy storage and insulation and in hormone formation. Lipids are organic macromolecules containing mostly carbon atoms, as well as hydrogen and oxygen atoms. Lipid monomers are fatty acids. Saturated fatty acids are saturated with hydrogen because their carbon atoms form only single bonds. As a result, saturated fats are solid at room temperature. In contrast, unsaturated fatty acids have gaps in their hydrogen saturation, because their carbon atoms form one or more double bonds. As a result, unsaturated fats are liquid at room temperature. Lipid polymers are called triglycerides. Saturated triglycerides contain only saturated fatty acids. Triglycerides are called unsaturated if they contain any unsaturated fatty acids.
NSV16036
64
views
Biology Quiz: What energy-carrying molecule is produced during cellular respiration?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
4
views
Carbohydrates
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#carbohydrates #saccharides #polysaccharides
SCIENCE ANIMATION TRANSCRIPT: In this video, we'll discuss carbohydrates. A carbohydrate is an organic macromolecule, which means it's a large molecule containing the element carbon. The hydrate part of carbohydrates means it also contains the components of water, hydrogen and oxygen. Carbohydrates always contain these elements in a proportion of one carbon atom, to two hydrogen atoms, to one oxygen atom. Think of it this way. All carbohydrate molecules will always have an equal number of carbon and oxygen atoms and will also always have twice as many hydrogen atoms. So why are carbohydrates important? Carbohydrates are the main fuel source from which all living things get their energy, but carbohydrates don't always have the same overall structure. They are classified as either simple or complex based on their chemical structure. All simple carbohydrates are sugars, referred to as saccharides. A saccharide can be made of one sugar molecule, called a monosaccharide, or two sugar molecules, called a disaccharide. An example of a monosaccharide is the sugar glucose. It's a monosaccharide because it's made of a single sugar molecule. Simple sugars like glucose are the quickest form of energy because your cells can break them down easily. Words ending in O-S-E or OSE are typically sugars, such as the monosaccharides fructose, galactose, and dextrose. Like glucose, these monosaccharides are all simple sugars because they are made up of a single sugar molecule. When glucose and fructose are chemically bonded together, we get a disaccharide called sucrose, commonly known as table sugar. Sucrose is a disaccharide because it's made up of two monosaccharides. Like monosaccharides, disaccharides such as sucrose also end in O-S-E because they're sugar molecules. Now let's talk about complex carbohydrates, also known as polysaccharides. Polysaccharides are very long chains of multiple monosaccharides chemically bonded together. An entire polysaccharide is referred to as a polymer, while each individual monosaccharide in this long chain is referred to as a monomer. Examples of polysaccharides include cellulose, starches, and glycogen. Cellulose is found in all plants. It's a structural carbohydrate that is found in the cell wall of every plant cell. Starches are found in many of the foods we eat, such as potatoes and corn. Starches are also found in grains such as wheat and rice. The body's digestive system breaks down starches into glucose molecules to fuel the activities of all of our cells. Extra glucose molecules that the body doesn't immediately need for energy are stored as polysaccharides called glycogen either in the liver or in skeletal muscles. To sum up, carbohydrates are organic macromolecules containing carbon, hydrogen, and oxygen in a one to two to one proportion. Carbohydrates are the main source of energy in living organisms. Carbohydrate monomers are simple sugars called monosaccharides. Disaccharides are two monosaccharides bonded together. Monosaccharide and disaccharide names typically end in OSE such as glucose, fructose, and sucrose. Carbohydrate polymers are called polysaccharides. Polysaccharides include cellulose, starches, and glycogen. [music]
NSV16030
26
views
Biology Quiz: What is the term for making ATP to provide energy for the cell?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
5
views
Biology Quiz: What organelle is the “powerhouse” of the cell?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
5
views
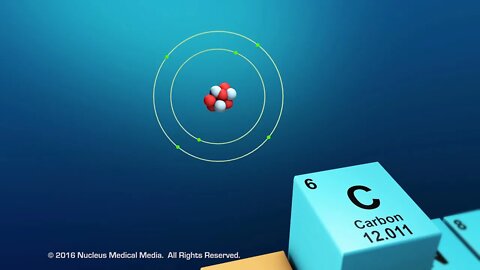
Overview of Organic Compounds
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#OrganicCompounds #OrganicMonemers #polymers
SCIENCE ANIMATION TRANSCRIPT: Today, we are going to be talking about organic compounds. Organic compounds are studied in biology because they are found in all living things. All organic compounds have the element carbon in them. In other words, all living organisms contain carbon. What's so special about the element, carbon? For one thing, no other element matches carbon's unique versatility to bond with other elements. Let's look at why this is true. For starters, carbon has an atomic number of six. That means that every atom of carbon has six protons in the nucleus. As an electrically neutral atom, carbon also has six electrons. Two core electrons are in the first energy level, which means it has four remaining valence electrons in the second energy level. Remember, valence electrons are those electrons available for bonding with other atoms. Accompanying these four electrons are four bonding sites, or four places that carbon can form bonds with other carbon atoms, or with atoms of other elements. Carbon's four valence electrons and four bonding sites allow it to form strong covalent bonds with many other elements, including hydrogen, oxygen, nitrogen, and phosphorus. Another feature of carbon atoms is they often form covalent bonds with other carbon atoms, to a nearly unlimited degree. This means that two carbon atoms can bond to one another, 50 carbon atoms can bond to one another, or even hundreds of carbon atoms can bond to one another. The ability of carbon atoms to bond to one another gives it the unique ability to shorten or lengthen a chain of carbon atoms to meet the very demands of the chemistry of life. So what kinds of molecules can carbon form? Well, small organic molecules called monomers are chemically bonded atoms that always include carbon. In addition to carbon, organic monomers usually contain hydrogen and oxygen, possibly along with nitrogen or phosphorus. Organic monomers often chemically bond to each other, joining together like beads on a string. This string of attached monomers will often continue to chemically bond with additional monomers, creating a much larger molecule, called a polymer. This process is called polymerization. Polymers may be made of different monomers or repeating units of the same monomer. Many organic polymers in the cells of living organisms are such large molecules that they're often referred to as macromolecules. DNA is an example of a macromolecule. Macro molecules can contain hundreds or even thousands of atoms. The four types of organic macromolecules are carbohydrates, lipids, proteins, and nucleic acids. Although they are all very large molecules, each type of organic macromolecule is distinct and different from the others. We'll discuss these four types of macromolecules in more detail separately. To sum up, organic compounds are found in all living things. All organic compounds contain the element carbon. Carbon atoms have a unique ability to bond to other carbon atoms, as well as other elements, such as hydrogen, oxygen, nitrogen, and phosphorus. Organic monomers are chemically bonded atoms that always include carbon. Polymerization is the process of creating long molecules, called polymers, from multiple bonded monomers. And macromolecules are very large organic molecules. [music]
NSV16027
46
views
Biology Quiz: Which organelles take in and break down cellular debris?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
8
views
Biology Quiz: Which organelle customizes proteins into forms the cell can use?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
11
views
pH
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#pH #HydrogenIons #HydroxideIons
SCIENCE ANIMATION TRANSCRIPT: Now that we've discussed acids and bases, let's talk about pH. The H in pH stands for hydrogen. pH is a measure of the concentration of free hydrogen ions in a solution. It tells us whether the solute in a solution is an acid or a base. Sometimes a base is referred to as being alkaline. To measure how acidic or basic a substance is, scientists use the pH scale, numbered from 1 to 14. The pH scale is an inverse scale of the concentration of hydrogen ions in a solution. The more hydrogen ions it contains, the lower its pH number and the stronger an acid it is. In contrast, the more hydroxide ions a solution has, the higher its pH number and the more basic or alkaline it is. Water is pH-neutral because it contains equal numbers of hydrogen and hydroxide ions. So water is right in the middle of the scale at a neutral pH of seven. Each whole number on the pH scale represents an exponential difference in value. This means that each whole number decrease in pH represents a 10 times increase in hydrogen ion concentration. For example, coffee has a pH of 5. So from a pH of 7 to a pH of 5, there is 10x10 or a 100-fold increase in hydrogen ion concentration compared to neutral water. As you can see, coffee is 100 times more acidic than water. This also means that coffee is 100 times less alkaline than water because it has a 100-fold decrease in hydroxide ion concentration. Baking soda is an example of a base with a pH of nine. It has a 100-fold decrease in hydrogen ion concentration compared to neutral water. However, it also has a 100 fold increase in hydroxide ion concentration. So baking soda is a hundred times more basic or alkaline than water. To function properly, biological processes in the human body require fluids with a pH in the range of approximately 6.5 to 7.5. For example, the normal pH of blood is 7.4. If acids or bases in the blood lower or raise the pH too much, a person could become sick or die. One way the body controls pH is through the use of different buffers. Buffers are substances that neutralize other acids and bases in solutions such as blood to prevent dramatic swings in pH. In summary, pH is a measure of the concentration of free hydrogen ions in a solution. A pH scale is an inverse scale of hydrogen ion concentration from 1 to 14. If the hydrogen ion and hydroxide ion concentrations are the same, the solution is pH-neutral. A pH of seven on the pH scale is neutral. Pure water has a pH of seven. Acids have a pH of less than seven and bases have a pH of greater than seven. The closer to seven a solution is, the weaker the acid or base. The farther away from seven a solution is, the stronger the acid or base. A buffer is a weak acid or base that reacts with a strong acid or base to prevent large changes in pH.
NSV16024
65
views
Biology Quiz: What sac-like organelle stores material within the cell?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
2
views